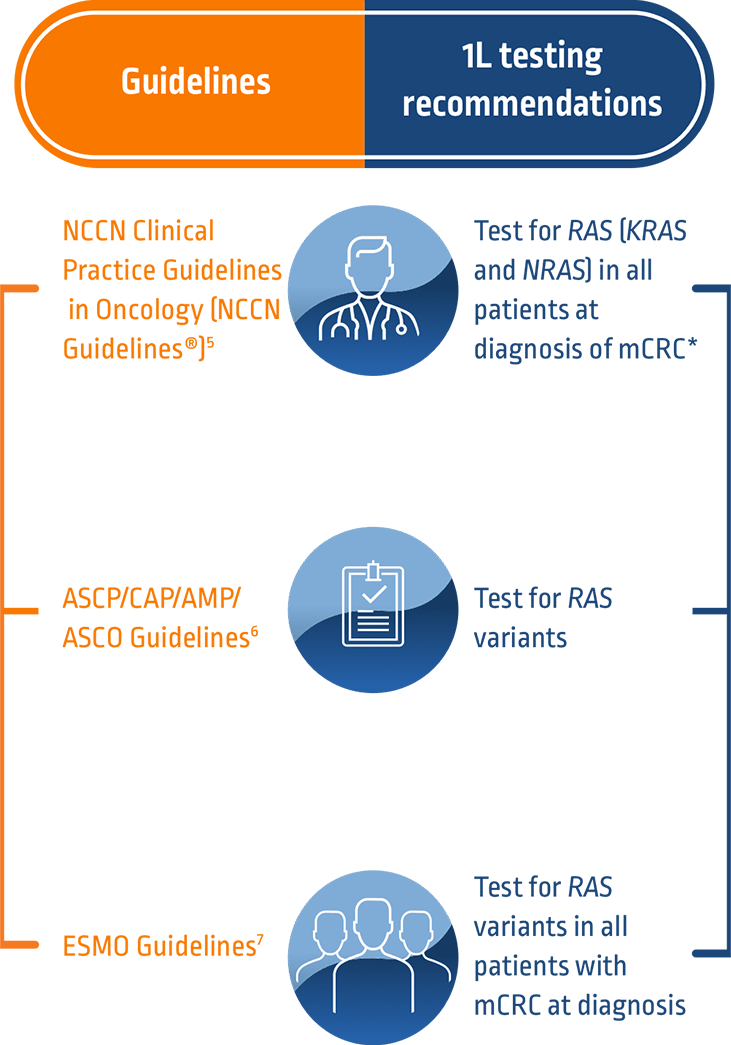
References: 1. Biller LH, et al. JAMA. 2021;325(7):669-685. 2. Cervantes A, et al. Ann Oncol. 2023;34(1):10-32. 3. Patel JN, et al. J Pers Med. 2019;9(1):3. 4. Taieb J, et al. Drugs. 2019;79(13):1375-1394. 5. Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Colon Cancer v.2.2023. © National Comprehensive Cancer Network, Inc. 2023. All rights reserved. Accessed April 25, 2023. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way. 6. Sepulveda AR, et al. J Mol Diagn. 2017;19(2):187-225. 7. ESMO Metastatic Colorectal Cancer Living Guideline. https://www.esmo.org/living-guidelines/esmo-metastatic-colorectal-cancer-living-guideline/diagnosis/article/diagnosis-pathology-and-molecular-biology. Accessed July 28, 2023. 8. NCCN Rectal. 9. Roche Diagnostics. https://diagnostics.roche.com/us/en/products/params/cobas-kras-mutation-test.html. Accessed June 9, 2023. 10. Conroy M, et al. Cancer Drug Resist. 2021;4(3):543-558. 11. Fakih M, et al. Oncologist. 2022;27(8):663-674.