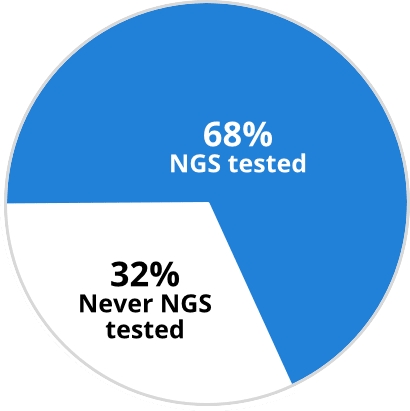
Why and who to test
Guidelines recognize the value of knowing KRAS G12C status in advanced NSCLC3-5
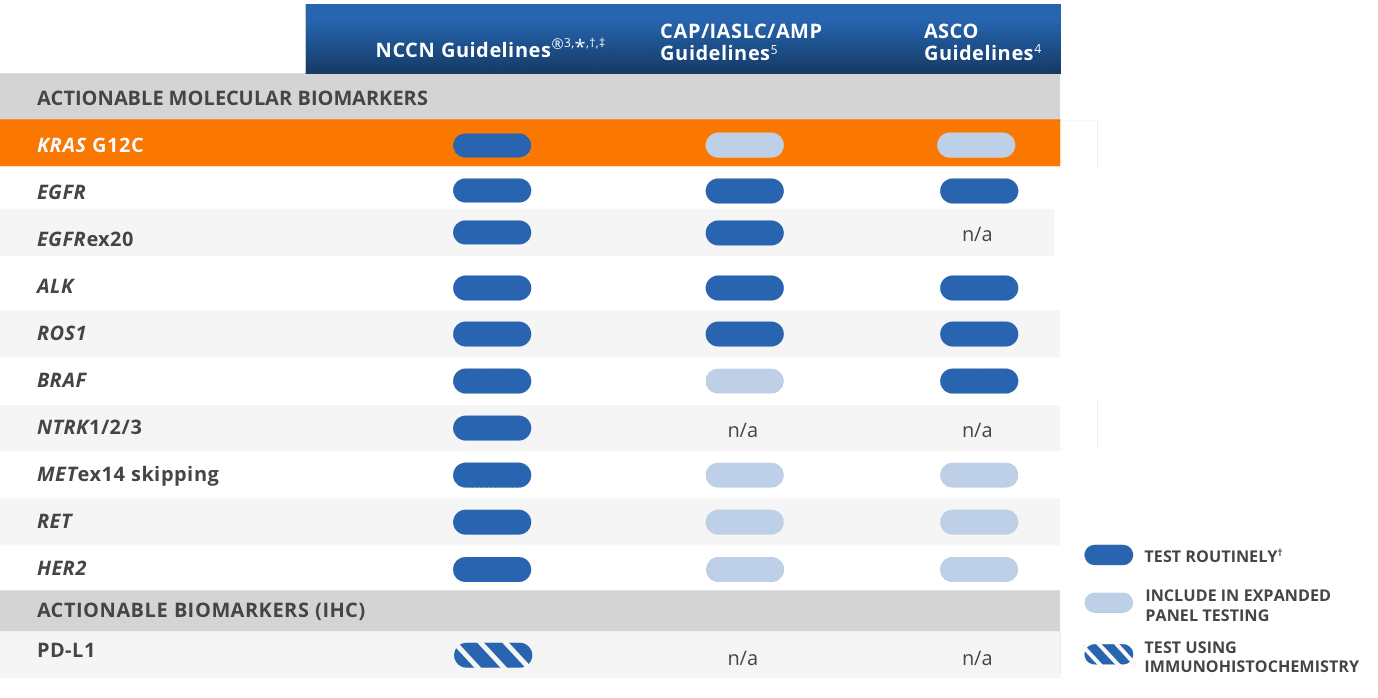

*Includes patients with advanced/metastatic NSCLC, but not locally advanced NSCLC.3
†It is recommended at this time that when feasible, testing be performed via a broad, panel-based approach, most typically performed by NGS.3
‡The NCCN Guidelines® for NSCLC provide recommendations for individual biomarkers that should be tested and recommend testing techniques but do not endorse any specific commercially available biomarker assays or commercial laboratories.3
Consider comprehensive genomic profiling at diagnosis to identify actionable biomarkers like KRAS G12C
- KRAS G12C is already included in most NGS panels and can also be detected by currently available PCR and Sanger sequencing methods7
Consider testing for KRAS G12C in eligible patients with NSCLC at diagnosis
How to test and report
The KRAS G12C mutation can be detected in tissue and liquid biopsy specimens using common molecular testing methods2
Most NGS platforms already test for KRAS G12C. There are also many PCR platforms capable of KRAS G12C testing7
- Below is a list of FDA-approved companion diagnostics (CDx) for KRAS G12C for patient identification in NSCLC12
| Test type | FDA-approved CDx for KRAS G12C patient identification |
|---|---|
| NGS |
|
| PCR | therascreen KRAS RGQ PCR kit (Qiagen Manchester, Ltd., tissue) |
- Other FDA-approved tests are available to test for KRAS G12C in NSCLC in addition to CDx tests. Reach out to your preferred reference lab for additional information
- CAP/IASLC/AMP Guidelines state that single-gene KRAS testing may be offered in addition to EGFR, ALK, and ROS1 to exclude patients from further testing5
- Consider listing KRAS G12C at the variant level in your reports, as some analytical reports may not specify this mutation13
Liquid biopsy may be used as an alternative to tissue-based testing14
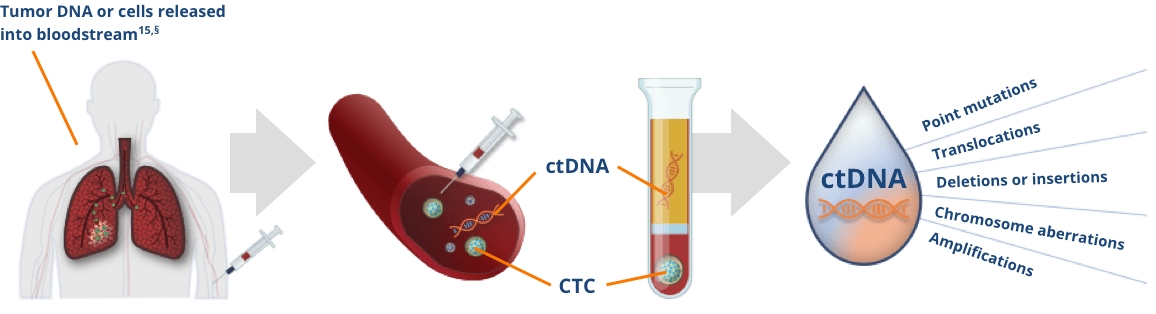
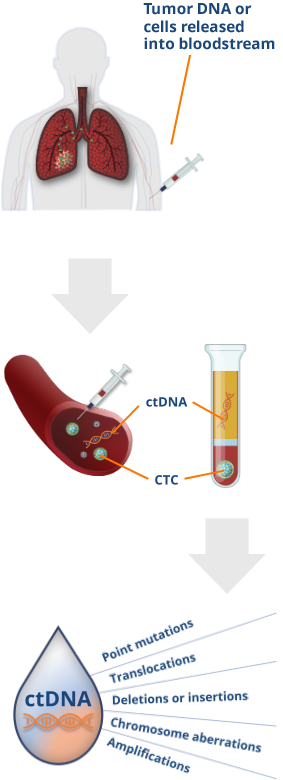
Image reproduced with permission from Pantel K, et al.15
§The total amount of ctDNA may be < 0.01% of the total cfDNA concentration.15
One form of liquid biopsy consists of obtaining ctDNA from plasma, which can be used to identify genomic alterations15
- Plasma from blood samples contains circulating cfDNA released from healthy cells and tumor cells
- Tumor cells release cfDNA known as ctDNA
Liquid biopsy can provide a comprehensive biomarker profile that may inform treatment decisions after 1L NSCLC therapy16,17
- Liquid biopsy may identify actionable mutations like KRAS G12C when tissue quantity is not sufficient16
- NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) state that cfDNA testing can be used when a patient is medically unfit for tissue biopsy and/or insufficient tissue is available for molecular analysis3
- The CAP/IASLC/AMP guidelines and an IASLC consensus paper support the utility of positive tissue and liquid biopsy results to inform treatment plan decisions16,18
- Because of variable tumor shedding and assay sensitivity, confirming negative liquid biopsy results with tissue biopsy whenever possible is recommended as mutations cannot be ruled out from a liquid sample alone16
- If tissue biopsy isn't an option for your patient, liquid biopsy may be an alternative16,18
Liquid biopsy can enable the identification of actionable biomarkers (such as KRAS G12C) and may inform treatment decisions following 1L NSCLC therapy16,17
- Liquid biopsy allows for detection of biomarkers when tissue is not available16
- In the NILE study, liquid biopsy had a faster median turnaround time of 9 days vs 15 days for tissue biopsy14,**
- Tissue biopsy in NSCLC remains the gold standard.16,17,21 However, using cfDNA in addition to tissue in the NILE study resulted in a 48% increase in the identification of patients with a guideline-recommended biomarker14,**
- If liquid biopsy results are negative, perform a tissue test when feasible as the presence of driver mutations cannot be ruled out from a liquid biopsy sample alone14,††
**Concordance of PPV for the other guideline-recommended biomarkers was similarly high.14
††Among 282 patients with untreated non-squamous mNSCLC, the NILE study (Non-invasive versus Invasive Lung Evaluation) (N=307) enrolled patients at one of 28 North American centers between July 2016 and April 2018. The number of patients identified to have a driver mutation increased from 60 to 89, including those with negative, not assessed for the biomarker identified in cfDNA, or insufficient tissue results. The guideline-recommended biomarkers in this study include: EGFR, ALK, ROS1, BRAF V600E, RET fusions, METex14 skipping, MET amplification, KRAS mutations, and ERBB2 (HER2). The number of patients who received complete genotyping were 268 cfDNA vs 51 tissue (P < 0.0001). Turnaround time was defined as the number of days between test order date and the retrieval of test results.14
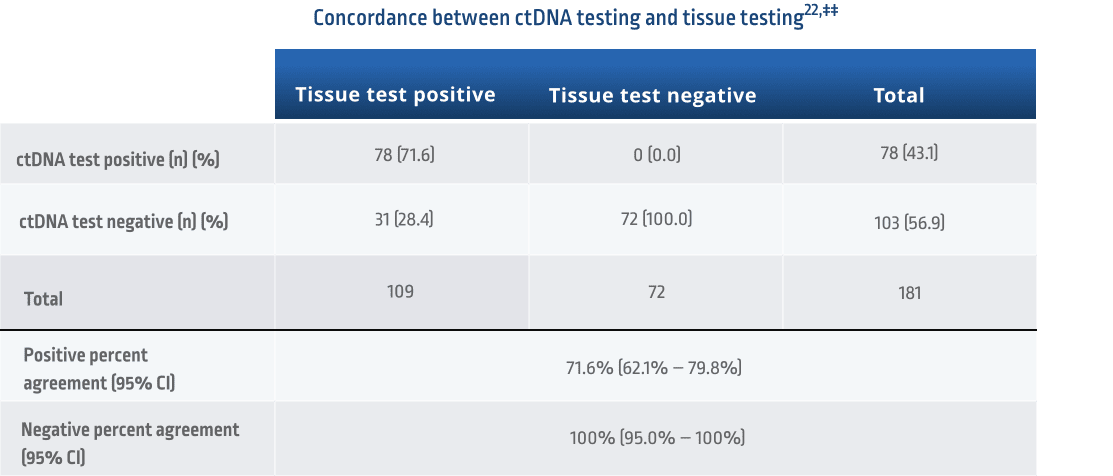
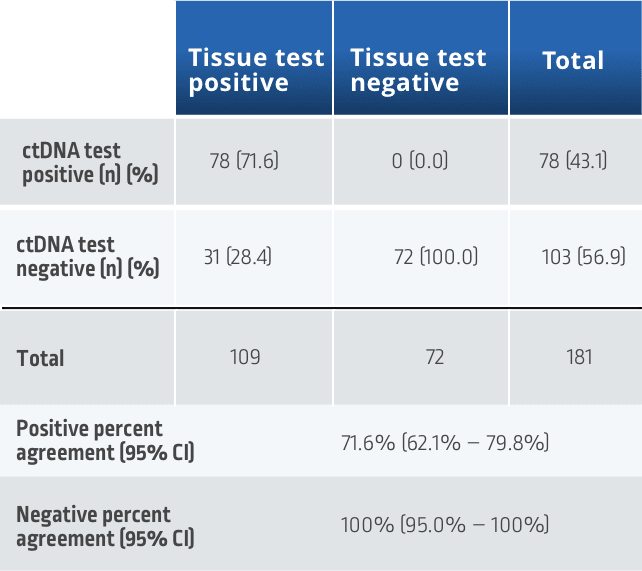
If a liquid biopsy returns a positive result, it is actionable. If results are negative for actionable biomarkers, it is recommended to perform a tissue test as the presence of driver mutations cannot be ruled out from a liquid biopsy sample alone16,18
‡‡Concordance measured using 109 matched plasma and tissue samples from the Amgen 20170543 study. Samples were evaluated for KRAS G12C-positive or -negative status using the therascreen KRAS RGQ PCR test for tissue and Guardant360 CDx for cfDNA.22
Comparing Tissue vs Liquid Biopsy NGS Testing
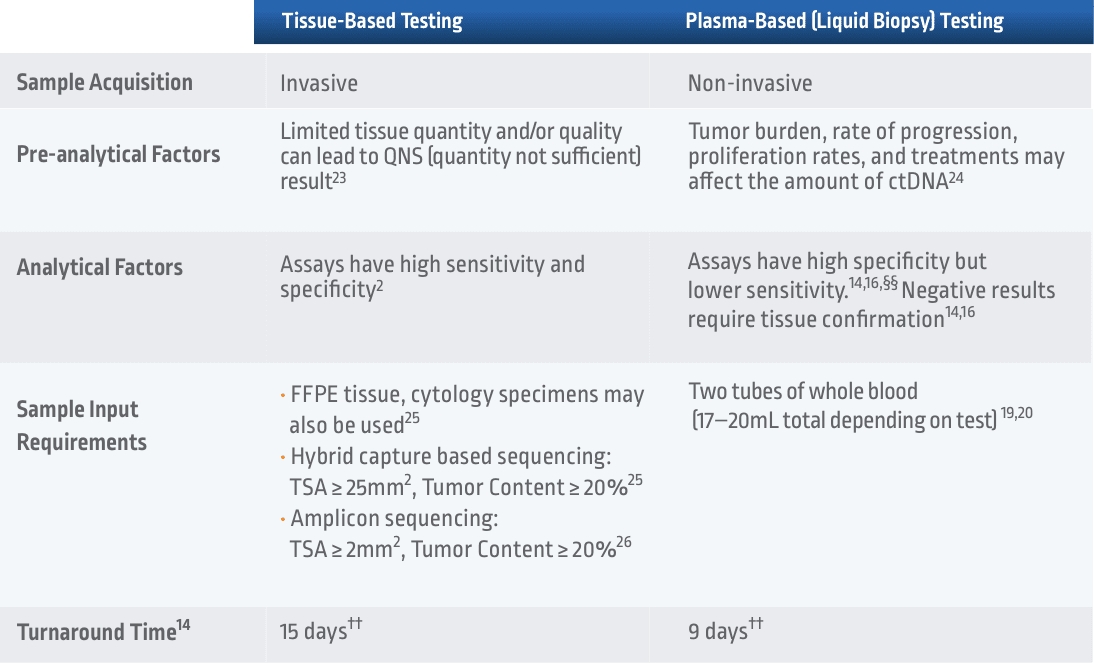
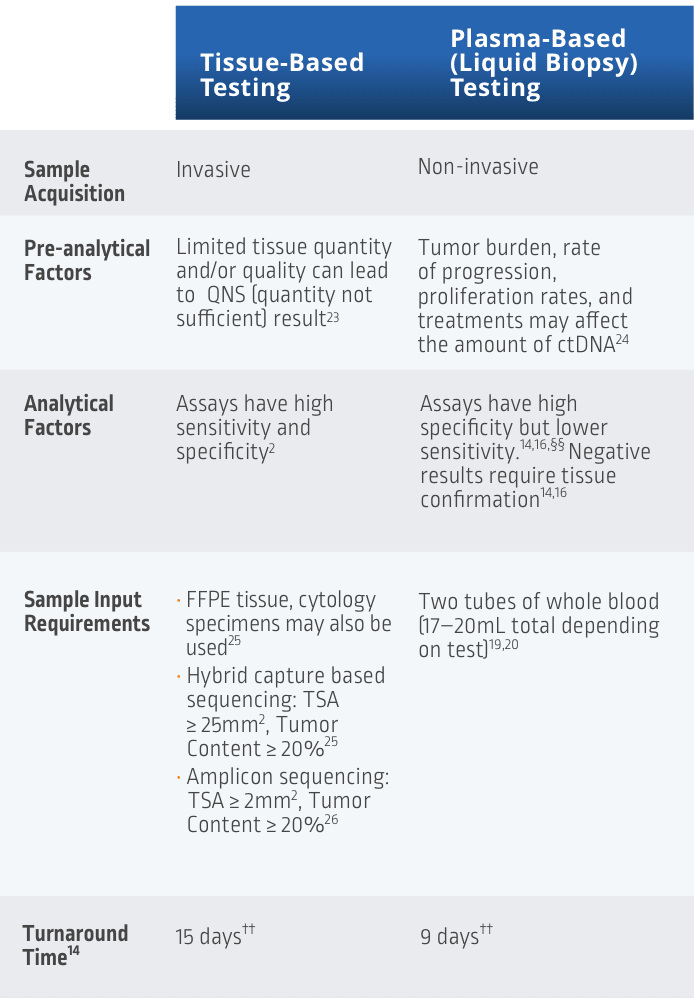
††Among 282 patients with untreated non-squamous mNSCLC, the NILE study (Non-invasive versus Invasive Lung Evaluation) (N=307) enrolled patients at one of 28 North American centers between July 2016 and April 2018. The number of patients identified to have a driver mutation increased from 60 to 89, including those with negative, not assessed for the biomarker identified in cfDNA, or insufficient tissue results. The guideline-recommended biomarkers in this study include: EGFR, ALK, ROS1, BRAF V600E, RET fusions, METex14 skipping, MET amplification, KRAS mutations, and ERBB2 (HER2). The number of patients who received complete genotyping were 268 cfDNA vs 51 tissue (P < 0.0001). Turnaround time was defined as the number of days between test order date and the retrieval of test results.14
§§FDA-approved targeted therapies included EGFR, ALK, ROS1, BRAF V600E, RET, MET exon 14, ERBB2 (HER2), and KRAS14,16
Reporting and documentation
Considerations that your multidisciplinary team (MDT) may find helpful when reporting biomarker results
Your MDT may include oncologists, pathologists, thoracic surgeons, pulmonologists, and others18
Where to test
The above is a list of facilities with high testing volume that are CLIA certified and accept external samples. CLIA certification was validated using the CDC website, and acceptance of external samples was confirmed by reviewing facility websites and/or contacting facilities directly. Amgen neither recommends nor endorses, and may or may not have financial relationships with, any facility that appears on this list. This list is not intended to be a comprehensive list nor as a referral to any lab listed. If you would like to suggest a facility to be added to this list, please contact Amgen MedInfo at 800-77-AMGEN.
This information is current as of September 9, 2021. Amgen does not guarantee the accuracy of this information, and it is up to the individual healthcare professional to conduct his/her own research.
1L, first line; ALK, anaplastic lymphoma kinase; AMP, Association for Molecular Pathology; ASCO, American Society of Clinical Oncology; BRAF, proto-oncogene B-Raf; CAP, College of American Pathologists; CDC, Centers for Disease Control and Prevention; CDx, companion diagnostic; cfDNA, circulating free DNA; CI, confidence interval; CLIA, Clinical Laboratory Improvement Amendments; CTC, circulating tumor cells; ctDNA, circulating tumor DNA; EGFR, epidermal growth factor receptor; EMR, electronic medical record; ERBB2, erb-B2 receptor tyrosine kinase 2; FFPE, formalin-fixed, paraffin-embedded; GGT, glycine; HER2, human epidermal growth factor receptor 2; IASLC, International Association for the Study of Lung Cancer; IHC, immunohistochemistry; KRAS, Kirsten rat sarcoma; MET, mesenchymal-to-epithelial transition; mNSCLC, metastatic non-small cell lung cancer; n/a, not applicable; NCCN, National Comprehensive Cancer Network; NGS, next-generation sequencing; NSCLC, non-small cell lung cancer; NTRK, neurotrophic tyrosine receptor kinase; PCR, polymerase chain reaction; PD-L1, programmed cell death ligand 1; PPV, positive predictive value; RET, rearranged during transfection; ROS1, c-ros oncogene 1; TGT, cysteine; TSA, tumor surface area.
References: 1. Data on file, Amgen; [Analysis of AACR Genie v12]. 2. Pennell NA, et al. Am Soc Clin Oncol Educ Book. 2019;39:531-542. 3. Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Non-Small Cell Lung Cancer v.2.2023. © National Comprehensive Cancer Network, Inc. 2023. All rights reserved. Accessed February 17, 2023. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way. 4. Kalemkerian GP, et al. J Clin Oncol. 2018;36:911-919. 5. Lindeman NI, et al. Arch Pathol Lab Med. 2018;142:321-346. 6. Pennell NA, et al. JCO Precis Oncol. 2019;3:1-9. 7. Sherwood JL, et al. ESMO Open. 2017;2:e000235. 8. Spira AI, et al. Lung Cancer. 2021;159:1-9. 9. Hess LM, et al. JTO Clin Res Rep. 2022;3:100336. 10. Mileham KF, et al. Presented at: The American Society of Clinical Oncology Annual Meeting; June 2022; Virtual Congress. 11. Bruno DS, et al. JCO Precis Oncol. 2022;6:e2100427. 12. FDA.gov. https://www.fda.gov/medical-devices/in-vitro-diagnostics/list-cleared-or-approved-companion-diagnostic-devices-in-vitro-and-imaging-tools. Accessed May 5, 2023. 13. Li MM, et al. J Mol Diagn. 2017;19:4-23. 14. Leighl NB, et al. Clin Cancer Res. 2019;25:4691-4700. 15. Pantel K, et al. Nat Rev Clin Oncol. 2019;16:409-424. 16. Rolfo C, et al. J Thorac Oncol. 2021. doi:10.1016/j.jtho.2021.06.017. 17. Rolfo C, et al. J Thorac Oncol. 2018;13:1248-1268. 18. Gregg JP, et al. Transl Lung Cancer Res. 2019;8:286-301. 19. FoundationOne® Liquid CDx. https://assets.ctfassets.net/w98cd481qyp0/3a8jFw3KUjIU3RWPdcT9Ax/dcb2ffd6d8d9a40a65ccf663269cc39a/FoundationOne_Liquid_CDx_Label_Technical_Info.pdf. Accessed September 10, 2021. 20. Guardant360 CDx. https://www.accessdata.fda.gov/cdrh_docs/pdf20/P200010A.pdf. Accessed October 6, 2021. 21. Tan AC et al. J Clin Oncol. 2022;40:611-625. 22. FDA.gov. https://www.accessdata.fda.gov/cdrh_docs/pdf20/P200010S002B.pdf. Accessed April 28, 2023. 23. Ascierto P, et al. J Mol Diagn. 2019;21:756-767. 24. Bronkhorst AJ, et al. Biomol Detect Quantif. 2019;17:100087. 25. FoundationOne® CDx. https://assets.ctfassets.net/w98cd481qyp0/6qYLg8jUuEYEvUytoBz8p6/f7764c8e3fcadda9ec1374fe26bb999e/F1CDx_Specimen_Instructions.pdf. Accessed May 11, 2023. 26. Tomlins SA, et al. JCO Precis Oncol. 2021;5:1312-1324. 27. Baskovitch BW, et al. College of American Pathologists. https://documents.cap.org/protocols/PleuraPericard_4.1.0.0.REL_CAPCP.pdf. Accessed April 12, 2023. 28. QIAGEN. https://www.qiagen.com/us/resources/download.aspx?id=59192bf9-d987-4de9-9130-649a87666d38&lang=en. Accessed September 9, 2021. 29. Kim ES, et al. J Thorac Oncol. 2019;14:338-342.