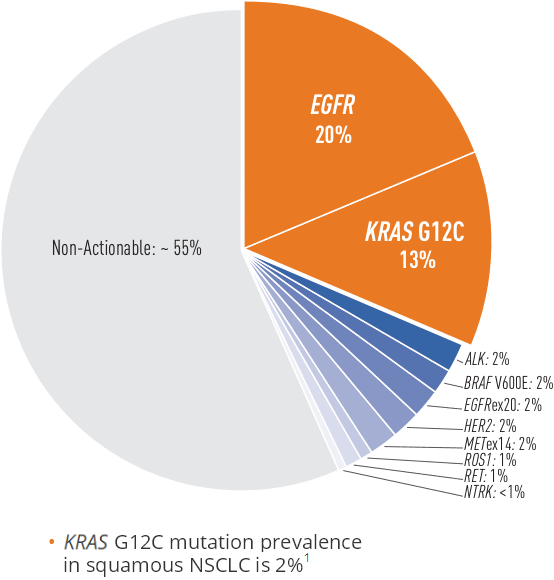
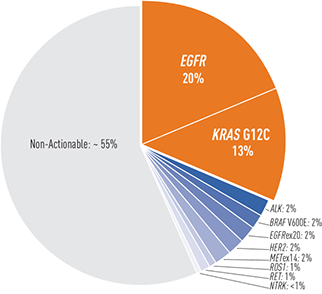
Prevalence of oncogenic drivers in non-squamous NSCLC1,*
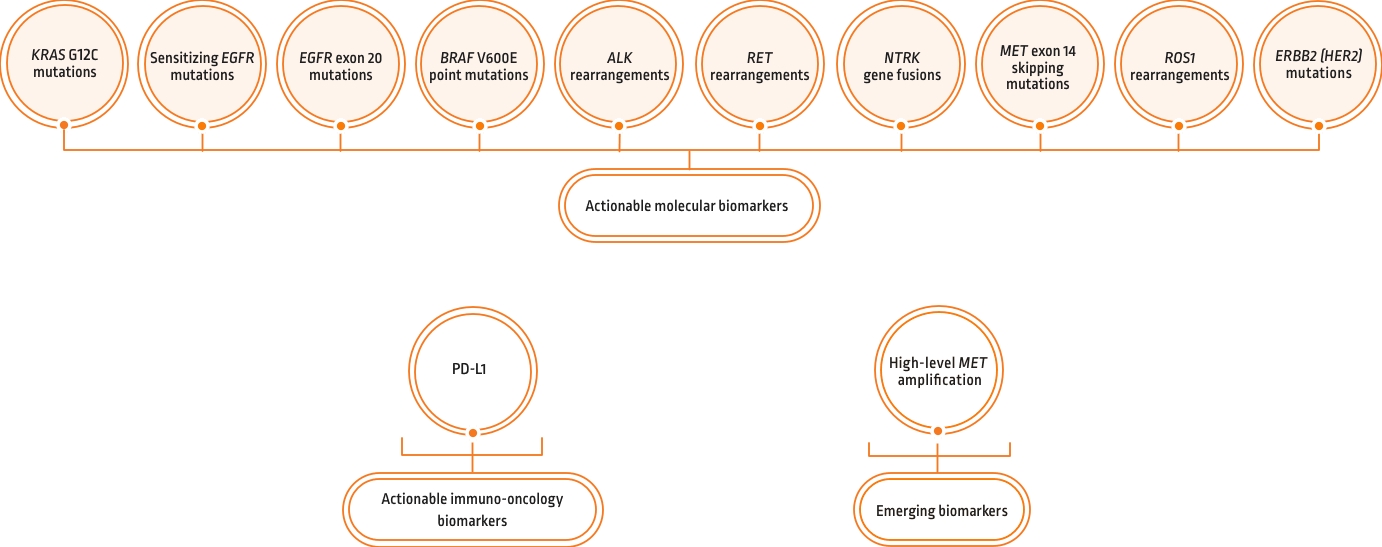
- KRAS G12C is one of the most common molecular biomarkers that can occur in any patient with NSCLC1
- KRAS mutations generally occur early and persist as the disease progresses8
- Since the KRAS G12C mutation rarely co-occurs with other actionable driver mutations, patients are unlikely to be eligible for therapies targeting these specific mutations (eg, EGFR, ALK, ROS1, BRAF)9
- Patients with advanced KRAS G12C–mutated NSCLC are more likely to be smokers, have non-squamous histology, and harbor more STK11 and KEAP1 co-mutations than the overall NSCLC population10
- Many patients do not receive comprehensive molecular testing11
- NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) recommend molecular testing, including testing for the KRAS G12C mutation, in eligible patients with advanced NSCLC7
KRAS G12C mutations can occur regardless of patient characteristics; therefore, consider testing for KRAS G12C in all eligible patients with NSCLC at diagnosis10
AACR, American Association for Cancer Research; ALK, anaplastic lymphoma kinase; BRAF, proto-oncogene B-Raf; EGFR, epidermal growth factor receptor; ERBB2, erb-B2 receptor tyrosine kinase 2; HER2, human epidermal growth factor receptor 2; KEAP1, kelch-like ECH-associated protein; KRAS, Kirsten rat sarcoma; MET, mesenchymal-to-epithelial transition; NCCN, National Comprehensive Cancer Network; NSCLC, non-small cell lung cancer; NTRK, neurotrophic tyrosine receptor kinase; PD-L1, programmed cell death ligand 1; RET, rearranged during transfection; ROS1, c-ros oncogene 1; STK11, serine/threonine kinase 11.
References: 1. Data on file, Amgen; [Analysis of AACR Genie v12]. 2. Tan AC, et al. J Clin Oncol. 2022;40:611-625. 3. Pennell NA, et al. Am Soc Clin Oncol Educ Book. 2019;39:531-542. 4. Mullard A, et al. Nat Rev Drug Discov. 2019;18:887-891. 5. Hess LM, et al. JTO Clin Res Rep. 2022;3:100336. 6. American Cancer Society. https://www.cancer.org/cancer/lung-cancer/treating-non-small-cell/targeted-therapies.html. Accessed April 1, 2023. 7. Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Non-Small Cell Lung Cancer v.2.2023. © National Comprehensive Cancer Network, Inc. 2023. All rights reserved. Accessed February 17, 2023. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way. 8. Thein KZ, et al. JCO Precis Oncol. 2022;6:e2100547. 9. Skoulidis F, et al. Nat Rev Cancer. 2019;19:495-509. 10. Spira A, et al. Lung Cancer. 2021;159:1-9. 11. Nadler E, et al. Presented at: The American Society of Clinical Oncology Annual Meeting; June 2021; Virtual Congress.